|
The left structure shows an oxygen atom with three lone pairs of electrons single bonded to a nitrogen atom with one lone pair of electrons that is double bonded to an oxygen with two lone pairs of electrons. Now N has 4 bonds and no lone pairs, so it owns 4 electrons. For the ammonium ion, NH 4 +, each H is still 0. Remember, electron counting to determine an octet counts all of the bonding and nonbonding electrons equally. This challenge of determining accurate atomic reference energies should be kept in mind, when molecular binding energies are considered. 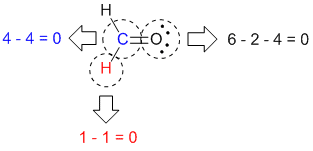
If it is two short, it has a two plus charge. This is good, because all the formal charges of each atom must add up to the total charge on the molecule or ion. the formal charge is additive: if the atom has two extra electrons in the molecule, it has a two minus charge. Now, lets look at the cationic form of methanol, CH3OH2+. (1 valence electron on isolated atom) - (0 nonbonding electrons) - (½ x 2 bonding electrons) 1 - 0 - 1 0. The electrons involved in the N–O double bond, however, are in different positions: For each H atom, it has 1 bond and thus 1 electron, so its formal charge is also 0. For each of the hydrogens in methanol, we also get a formal charge of zero: formal charge on hydrogen. But for the octet rule I count the bond as 2 electrons (total of 8 electrons), so I am confused. Abstract: The heterogeneous electron transfer rate at a mercury-dimethylformamide interface was determined by ac polar. The sum of the formal charges of all the atoms equals 1, which is identical to the charge of the ion (1). In the book formal charge is calculated by adding up the electrons where each bond counts as 1 electron, so in the image for the oxygen with a formal charge of -1, we get a total of 7 electrons which is greater than the 6 that oxygen originally has. In this example, the nitrogen and each hydrogen has a formal charge of zero.\) can have two possible structures with the atoms in the same positions. Subtract this number from the number of valence electrons for the neutral atom: I: 7 8 1. 1) F o r m a l c h a r g e V a l e n c e e l e c t r o n s n.  
It can be obtained through: Formal charge Valence electrons no bonding electrons bonding electrons 2 (Eq. See this post of the nitrate resonance structures. atom) (cationic charge) + (anionic charge). Formal charge varies when you look at resonance structure. It plays a vital role in determining the shape of the molecule. Formal charge number of electrons an atom has in its natural state number of. Formal charge is the charge of an atom in a molecule. The sum of the formal charges of each atom must be equal to the overall charge of the molecule or ion. structures, choose the one with the best formal charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
